By Julia Pian, Senior Associate at Atlas Venture, as part of the From The Trenches features of LifeSciVC
As Jonathan Larson beautifully explores in “Seasons of Love” from Rent, it’s almost impossible to find the right variables to capture life experiences. Yet frequently in the diligence process, we are trying to understand whether indications like ADA-SCID or CGD have “enough” treatable patients or indications like Heme B or CF have “enough” remaining unmet need given existing therapies to garner venture investment in the early R&D process.
In this post, we will walk through how one integrates key aspects of the patient population and demographic, as well as the patient experience into assessment of an opportunity, and show how defining clinical unmet need goes hand in hand with developing your clinical development plan, regulatory strategy, and ultimately market access and reimbursement strategy. And, finally, we’ll answer the question if there is a peak sales number required for an Atlas investment (spoiler alert: no!). This piece will approach these questions through a rare disease lens, although similar principles can be applied to answering whether a prevalent but therapeutically crowded indication has enough “unmet need” to support an “n-th” therapeutic option.
I want to be clear that one can could never truly reduce a patient’s experience of their disease or the burden of a disease on a population to a number. Why then does it appear to be that we are trying to do just that? The people of the biopharma ecosystem are working to improve patients’ lives and outcomes. However, we are ultimately in an industry that requires large aliquots of capital to move life-changing science forward to patients. A recent analysis from the London School of Economics estimates the median research and development cost per approved product is $1.1B1. The capital intensity of the biopharma industry makes financial incentives a necessary aspect to create a sustainable flow of innovation for patients2.
At Atlas, we aim to “do well by doing good”, in other words, build and back companies that create medicines with positive impact, and let the financial returns follow. Therefore, understanding just how much we can do for patients becomes paramount! People are sometimes surprised to find that we are not routinely building discounted cash flow revenue models for programs that we’re evaluating. Frankly, we know it is impossible to prognosticate perfectly, especially at the earliest stages of development, and aim to avoid overconfidence and false precision. Many drugs have proven even the best Wall Street analysts (let alone early-stage investors) wrong by underperforming or overperforming their sales estimates.
A decade ago, ATTR-CM was thought to be a rare disease with perhaps 200K patients worldwide (Ionis Corporate Materials, 2015), but today estimates place the market at a size of 200K patients in the U.S. alone (and >500K globally). The emergence of novel, effective therapies can have profound impacts on clinical diagnosis practices and expand the market. On the other hand, there were strong hopes for Casgevy from VRTX/CRSP given transformative efficacy results. One analyst in 2020 predicted ~$400M sales in 2025. However, actual sales of Casgevy in 2025 ended up at a much more modest $115.8M, despite higher pricing than anticipated, potentially due to complexities of delivering the medicine and perhaps a different risk-benefit calculus from patients and physicians than originally anticipated.
Overall, we proceed with caution and do our best to understand the patient journey through care, and what opportunity exists for improving that care to help guide early investment and development decisions.
Beyond indication selection: hitting the bullseye with your target patient population
Highly efficacious, blockbuster drugs often treat only single digit percentages of patients with a given disease. Although market penetration is typically higher in rare disease, defining the phenotype of patient that will be the ideal candidate for your therapy is still key.
One way to begin to refine your target patient population is by imagining Figure 1 of your future NEJM paper declaring clinical proof of concept. I like to call this productive daydreaming! Figure 1 shares the demographic background of the patients of the trial, often divided by placebo and various treatment groups, and including such rows as time since diagnosis, number of prior lines of therapy, or baseline biomarker values or clinical scores. A non-exhaustive list of questions to consider is included below.
- What segment of the population can you treat?
- Will the risk-benefit of your potential therapy allow it to address patients with mild, moderate, and/or severe disease?
- Can you identify patients that may respond to your therapy by any of the following
- Genetics
- Biomarkers
- Clinical Symptoms
- Demographics
- Are there frequent comorbidities or complications of the disease that might preclude treatment with your therapy?
- Are there preferences around frequency and route of administration that might preclude patients from seeking out your therapy (ranging from needlephobia to requirement for an invasive procedure like ICV administration)?
- Where in the patient journey would your medicine fit in?
- How long since patients have been diagnosed would they start treatment?
- What is the existing standard of care?
- How many lines of therapy exist?
- What is first line treatment and what percentage fail and move on to second line? Third? Etc.
- What percentage of patients benefit from the therapy?
- How much do patients that respond benefit?
- Are there significant side effects or toxicities that are burden to patients? What percentage of patients experience those?
- Do patients have significant issues accessing the current standard of care?
- What line of therapy would your work come into market as?
- Are there competitors in clinical trial that will hit the market in the coming few years? How will that change the standard of care?
- Are there patients who have progressed to a point where your mechanism may not be efficacious?
- Can you actually reach your target patient population?
- What portions of patients are even connected to the medical system for their disease or have frequent touch points with a medical provider?
- Where are these addressable patients geographically located? Estimates of patient numbers often break-out US vs. ex-US patient numbers due to current drug pricing dynamics.
- What is the breakdown of insurance coverage in your target patient population?
- How large of a salesforce might be needed to reach the population?
- Is patient care concentrated in academic or tertiary care centers or, on the other hand, would primary care providers be the main prescriber base?
There are many potential ways to visualize this target population with one commercial-grade example below from recently acquired Atlas portfolio company Day One Biopharma. This figure shows, on the left, the specific pLGG patients addressable first-line, and, on the right, utilizes progression rates to help define the prevalent 2L+ pLGG patients that Ojemda is currently approved for. In this way, they are capturing both the current addressable market and potential expansion market in one slide.
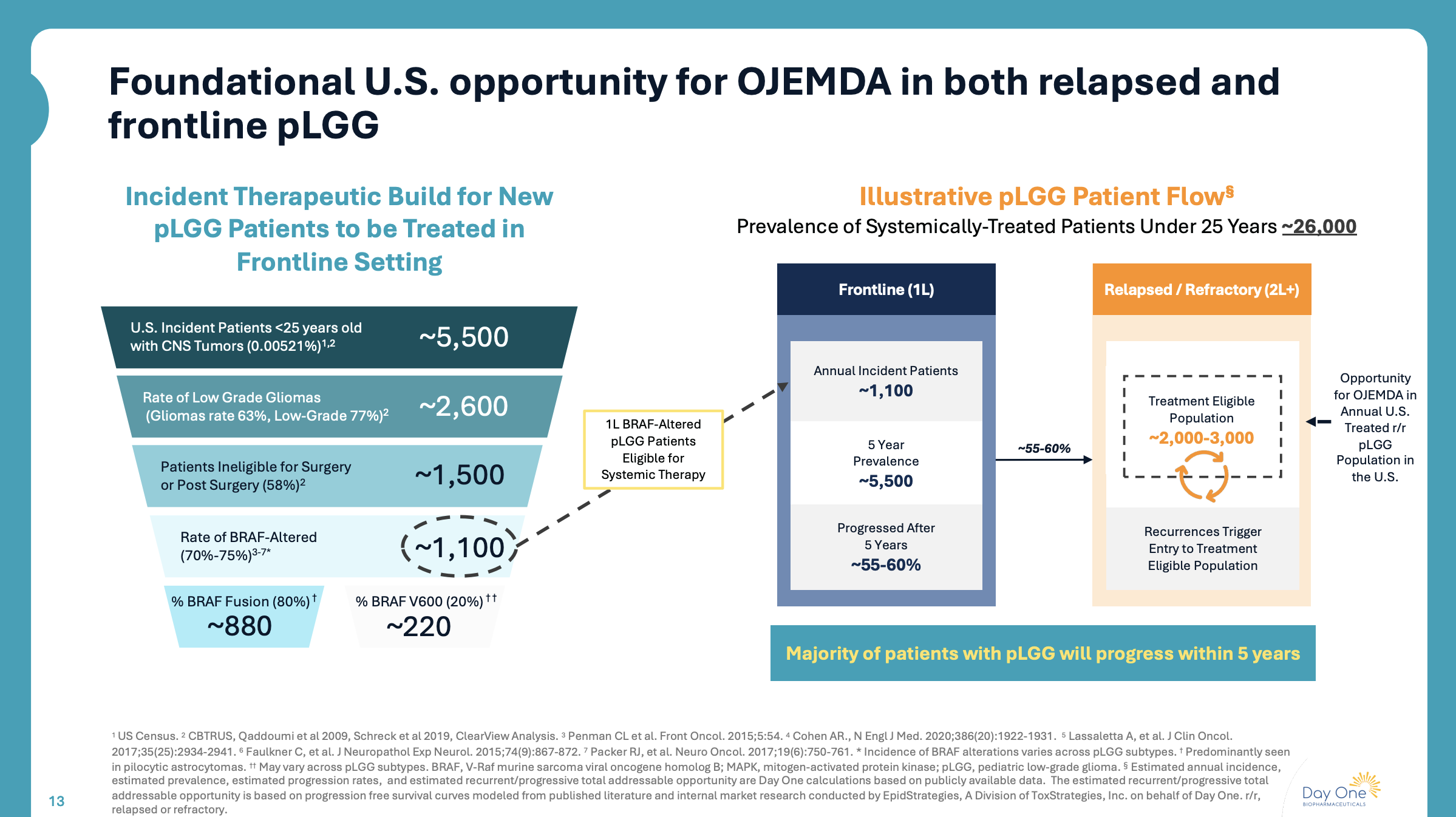
Figure 1. Visualizing your target patient population (Source: Day One Biopharma, March 2026)
Quantifying the Burden of Disease
Once you have identified your target patient population, the next step is to better understand the burden of disease. There are established quantitative frameworks, particularly Quality Adjusted Life Years (QALYs), Disability Adjusted Life Years (DALYs), and equal value Life Year (evLY), that Health Technology Assessment (HTA) agencies such as NICE and ICER often use as a standardized assessment of the cost effectiveness of a potential therapy. Although useful for cross therapy and indication comparisons, like any metric that aims to be broadly applicable, QALYs and associated measures are by nature reductive and do not fully capture what quality means for a given patient population, the waxing and waning nature of some illnesses, and spillover burden outside the patient.
Even for patients that share disease pathophysiology at the cellular level—an altered nucleotide or amino acid, perhaps—the physical, mental, and social effects of their disease is unique. The following questions are a starting point to better understand a patient’s healthcare experiences and defining the healthcare costs associated with living with a particular illness:
- How often are patients interacting with the healthcare system
- How many doctor’s visits do patients have per year?
- Which specialists do they see and how frequently do they see each?
- How often are patients getting laboratory tests or imaging to monitor their disease
- How many urgent care or emergency department visits occur per year
- How often are patients hospitalized?
- How long is a typical hospitalization? Is a stay at a rehabilitation center required after?
- What are the most common medical complications of a disease?
- A similar, but separate question: What are patients’ most feared complication of the disease?
- What is a patient’s life like outside of doctor’s visits and hospitalizations?
- How frequently are patients experiencing symptoms of their disease?
- What are the most frequent symptoms?
- What symptom is most burdensome?
- What is the impact on a patient’s ability to perform ADLs/IADLs?
- What other aspects of life are limited by disease (work, hobbies, socializing, eating, going to the bathroom, etc.)?
- Are there caregiver needs associated with this illness?
- How much do those needs typically cost?
- Is there lost productivity for caregivers, including time-off work required?
- Are there mental health impacts on caregivers?
- How many medications are they taking on average?
- What frequency and route of administration are these medications?
- Do treatments come with monitoring burdens, including laboratory testing?
- What can you learn from previous work done in the indication?
- How well is the standard of care addressing patient’s needs?
- Have there been regulatory submissions or HTA work in your chosen indication?
- What can you learn from those, including variables used to quantify burden of disease or areas that matter most to regulators and HTA organizations?
- How many doctor’s visits do patients have per year?
One particularly concrete and valuable example of putting in the work to understand your target indication comes from Atlas portfolio company Diagonal Therapeutics, which is advancing an ALK1 activating antibody for Hereditary Hemorrhagic Telangiectasia (HHT), a genetic bleeding disorder with no approved therapies and limited clinical development in the space to date. Diagonal collaborated with one of the leading key opinion leaders and patient advocacy organizations in the field to utilize the Komodo Healthcare Map claims database to quantify the mean patient per year healthcare costs for those living with HHT. This work was published in the American Journal of Hematology and SVP, Finance and Corporate Development, Eric Duhaime, was the senior author on the analysis. They found that for patients that have anemia and receive hematological support, such as iron infusions and/or red blood cell transfusions, annual healthcare costs were $40,000, greater than the average healthcare costs incurred by patients with sickle cell disease or muscular dystrophy and just under the $41,680 of healthcare costs incurred by patients with cystic fibrosis. Since there was limited biopharma interaction with the HHT population in the past, grounding the amount of healthcare costs incurred to diseases that the industry is more familiar with, like SCD and CF, puts a spotlight on the severity of the unmet need in the space.
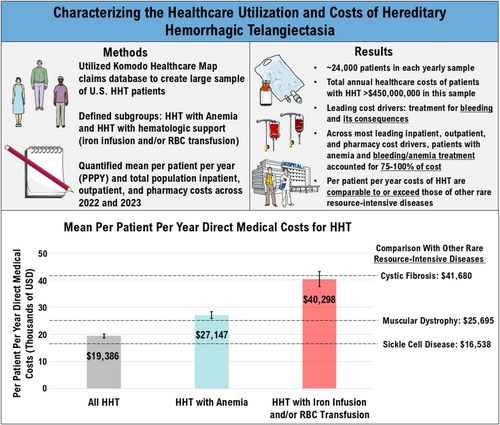
Figure 2. Graphical Abstract from Al-Samkari et al 2025 Am J Hematology
The importance of engaging patients, patient advocacy groups and physicians in this work early cannot be understated. This has been written about powerfully by many in the ecosystem including Ivana Magovcevic-Liebisch (former CEO of Vigil Neuroscience), Josh Brumm (former CEO of Dyne Therapeutics), and JC Gutierrez-Ramos (former CEO of Synlogic Therapeutics). Collaboration can and should go beyond KOL conversations or a Patient Impact Day, and longitudinal engagements with both physicians (on a Clinical Advisory Board or as investigators), and with patients perhaps through a natural history study (whether prospective, retrospective, or registry-based) are incredibly valuable. Natural history data is critical for understanding disease trajectory and for identifying the clinical milestones that matter most. A key part of development in a new indication may be collaborating with patients to define the very endpoints that may help the FDA ultimately decide if a drug helps patients “feel, function, or survive”3,4. Many rare diseases do not have a standardized scoring system à la EASI (atopic dermatitis) or SLEDAI (lupus), and establishing and validating these disease specific scoring systems can often happen almost in parallel with running the clinical program.
Thinking even further down the road, there is often a tremendous amount of market shaping work required when launching into a previously untreated indication, such as physician and patient education, introduction of novel diagnostics, or shifting established specialist referral patterns. This time-intensive and costly work is often put off by biotech teams as something that experienced large pharma commercial teams can take care of post-acquisition. However, upfront investment of time and money into building the case for unmet need may be a worthy investment, especially when you are working in an area where the commercial opportunity is not obvious.
How does unmet need factor into your thinking as a investor?
As a physician (particularly pediatrician) with a penchant for working to solve rare disease, I frequently come across entrepreneurs that ask what is the peak sales number that you need to see to consider investment? Or what’s the lowest prevalence disease that you’d consider in indication selection? Although you may hear about peak revenue cut-offs that pharma sets for potential acquisition targets, at least at Atlas, there is no hard and fast number that we use to consider an opportunity commercially viable. These estimates and numbers do not exist in a vacuum and instead one must consider the context around a potential program to better understand the investment opportunity.
Although stating the obvious, the metric that looms large in decision making for a venture capitalist is potential returns, and returns are made up of not only potential exit value, but also capital required to realize that exit value and probability of success. Smaller potential market sizes may still be viable if the capital required to get a drug approved and to patients or to a key value inflection point is relatively small and well-understood biology or precedented mechanisms increase the probability of success.
At the end of the day, generating numbers to describe patient’s unmet need is an important aspect of the process of developing a therapy. If you’ll humor some theater analogies once again, it’s shining a spotlight on patient’s experiences. However, it’s also important to not anchor on any given number, but to do our best to take in the whole stage, and consider the broader context before setting off on the sometimes dramatic and twisting journey that is drug discovery.
Acknowledgetments: Many thanks to Eric Duhaime, Alex Harding, Niranajan Kameswaran, and Essra Ridha for their time and feedback on this piece and many other projects!
Footnotes
- Bruce last wrote about this topic over a decade ago in 2014 where the Tufts Center for the Study of Drug Development named a price tag of $2.6B invested to drive one new approved drug. Although this seems unfathomably large, it held up to even a certain ex-McKinsey consultant’s scrutiny.
- I would be remiss in not acknowledging that the financial reality of the biopharma industry leads to patients with ultra-rare disease being underserved. When the number of patients with a given disease is so small, even regulatory incentives do not meaningfully shift the financial calculus and there can be limited allocation of resources to advancing novel therapies for those indications. There is great work ongoing to ensure that patients with ultra-rare disease can have access to life changing innovations (including by an organization I work with the N-of-1 Collaborative) and recent developments have reignited my hope that regulatory innovation by groups, including the US FDA and UK MHRA, will soon allow for sustainable funding of therapeutic development for patient populations down to a size of n=1.
- For instance, Medivation and academics collaborated on a study to understand which phenotypic traits caregivers of patients with Fragile X disease care most about. They found that controlling behavior and caring for oneself were ranked the highest and published their findings in the Journal of Developmental Behavioral Pediatrics. You may spot Atlas EIR, Judy Dunn, among the authors!
- The FDA actually has a series of guidance documents on Patient-Focused Drug Development and an ongoing effort in the Rare Disease Endpoint Advancement Pilot Program which we recommend for a deeper dive into these topics.
